Product name:2-Amino-4-methylthiazole-5-carboxylic acid
MF:C5H6N2O2S
MW: 158.18
Cas No.:67899-00-7
Structure: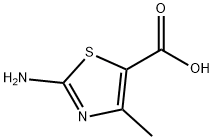
Appearance: Off-white solid
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
sodium hydroxide; ethanol;
Reactants are commercially availlable.
To a solution of ethyl 2-amino-4-methyl-1,3-thiazole-5-carboxylate 5 (3.00g, 16.1mmol) in ethanol (100mL) was added 1M sodium hydroxide solution (50.0mL) at room temperature, and the mixture was stirred for 26h. After the volatiles of the mixture were removed in vacuo, water and acetic acid were added to the residue to adjust the pH to 5.0. The resulting precipitate was collected by filtration, washed with water, and dried in vacuo to yield 6 (2.48g, 97percent) as a colorless powder. 1H NMR (200MHz, DMSO-d6) delta 2.35 (s, 3H), 7.57 (s, 2H); MS (ESI): m/z 159 [M+H]+, 157 [M?H].
