Product name:3-Bromo-6,7-dimethoxyquinoline
MF:C11H10BrNO2
Cas No.:850352-73-7
MW:268.1066
Structure: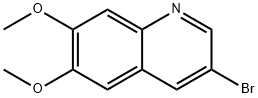
Purity:95%
Appearance:white solid
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
sodium hydroxide; water; hydrogen bromide; ethyl acetate; copper(I) bromide
Reactants are commercially availlable.
Wyeth, US2003/32645, A1, (2003) Step 5 8-Bromo-6-methoxyquinoline To 5.05 g (29.0 mmol) 6-methoxy-quinolin-8-yl-amine in 25 mL of 48 percent HBr at 0°C. is added a solution of 2.60 g (37.7 mmol) and 20 mL H2O. After stirring at 0°C . for 15 min, the resulting mixture is added dropwise to a 75°C. solution of 5.0 g (34.8 mmol) CuBr and 60 mL of 48percent HBr. After 5.5 h, the reaction mixture is neutralized with 150 mL of ice col d 5N NaOH, the resulting mixture is stirred with 300 mL EtOAc and filtered through a pad of celite. This mixture is extracted 2*100 mL EtOAc, and the combined organics are washed with 1*200 mL H2O, 1* 200 mL brine, dried over Na2SO4, filtered, and the volatiles are evaporated. Flash chromatography on SiO2 gel, eluding with hexanes/EtOAc (4/1 to 1/1), gives 4.23 g (17.8 mmol, a 61percent yield) of t he title compound as a brown oil. MS (ES) m/z 239 (MH)+.
