Product name:1-Fluoro-cyclopropanecarboxylic acid
MF:C4H5FO2
Cas No.:137081-41-5
MW:104.08
Structure: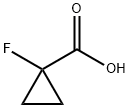
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
hydrogenchloride; sodium hydroxide; tert-butyl methyl ether; water;
Reactants can be synthesized in 1 step.
Bayer Aktiengesellschaft, US5434303, A1, (1995) b) Preparation of 1-fluoro-cyclopropane-1-carboxylic acid, of the formula STR16 A solution of 138 g (0.64 mol) of 4-chlorophenyl 1-fluoro-cyclopropane-1-carboxylate in 250 ml of methyl tert.-butyl et her is treated at 0°C. with a solution of 25.6 g (0.64 mol) of sodium hydroxide in 250 ml of water. The mixture is stirred for 3 hours at room temperature and subsequently extracted for 18 hours in a liquid-liquid extractor, using methyl tert.-butyl ether. The aqueous phase is acidified with 150 ml of concentrated hydrochloric acid, with ice-cooling, and then extracted three times with 300 ml por tions of diethyl ether. This organic phase is dried over magnesium sulphate and then concentrated by stripping off the solvent under reduced pressure. In this manner, 52 g (78percent of theory) of 1-f luoro-cyclopropane-1-carboxylic acid are obtained in the form of a solid substance of melting point 66°-68°C. 19 F NMR (188 MHz, CDCl3;CFCl3): delta=199 ppm 1 H NMR (200 MHz, CDCl3 /TMS): delta=1.40- 1.52 ppm (m, 4H, cyclopropyl H), 9.25 ppm (1H, COOH).
