Product name:2,4,6-TRIMETHYLPHENYLHYDRAZINE HYDROCHLORIDE
MF:C9H15ClN2
MW : 186.68
Cas No.: 76195-82-9
Structure: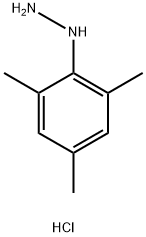
Purity:95%
Appearance:pink solid
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
hydrogenchloride; sodium nitrite; sodium hydroxide; tin(ll) chloride; water; water;
Reactants are commercially availlable.
THE REGENTS OF THE UNIVERSITY OF CALIFORNIA, WO2007/124494, A2, (2007) A 3.0 L, 3-neck round bottom flask equipped with a mechanical stirrer and an addition funnel was charged with concentrated hydrochloric acid (0.125 L) and deionized water ( 0.100 L ). The solution was cooled to -5 0C in an ice/salt water bath, and 2,4,6-trimethylaniline (70 niL, 0.50 mol, 1.0 equiv.) was added dropwise over 30 minutes; during this time a white precipitate formed. The mixture was s tirred for an additional 15-20 minutes before 52.5 mL of a 9.5 M aq. solution of sodium nitrite (70 mL, 0.50 mol, 1.0 equiv. ) was added dropwise over 30^45 minutes. The resulting brown solution was a llowed to stir for 30 minutes before a 3.3 M solution of stannous chloride dihydrate in 1 : 1 concentrated HChH2O (0.455 L, 1.5 mol, 2.5 equiv.) was added over 4 hours with vigorous stirring. (Upon ad dition of the stannous chloride solution a thick light orange slurry had formed which sometimes caused seizure of the mechanical stirrer. Addition small amounts of deionized water helped to facilitate stirring again.) The mixture was allowed to warm to room temperature and stirred vigorously for 16 hours. The mixture was cooled to 0 0C for 1 hour before filtering through a fine porosity glass frit ted buchner funnel, and the collected orange solid washed with brine and diethyl ether. This orange solid was added to a 2-L round-bottom charged with 10 M NaOH(aq) (1.0 L) and diethyl ether (0.700 L) and cooled to -5 0C. Once nearly all of the solid had dissolved (ca. 1 h), the organic layer was removed, and the aqueous layer extracted with diethyl ether (2 x 0.400 L). The combined organic fracti ons were dried over Na2SO4, filtered into a flame-dried 2.0 L round-bottom flask, placed under an atmosphere of N2(g), cooled to 0 0C, and treated with 4 M HCl in 1 ,4-dioxane (0.125 L). A white preci pitate formed, was collected by vacuum filtration, washed with diethyl ether, and recrystalized from 200 proof EtOH to afford the title compound (50 g, 0.27 mol, 54percent) as a white powder. 1H NMR ( 400 MHz, DMSO) delta 9.57 (3H, s, br, NH, varies), 6.89 (s, 2H), 2.33 (s, 6H), 2.21 (s, 3H); 13C NMR (100 MHz, DMSO) delta 138.06, 136.20, 135.01, 129.17, 20.54, 17.95; IR (thin film) v 3294 , 3002, 2964, 2911, 2680, 1564, 1513, 1481, 852, 825, 753 cm"1; HRESI+/TOF-MS calcd for C9Hi5N2+ [M]+ 151.1230, found 134.0921 for aniline anion radical (134.0975).
