Product name:1-Bromo-2-ethyl-6-methylbenzene
MF:C9H11Br
MW: 199.09
Cas No.:65232-55-5
Structure: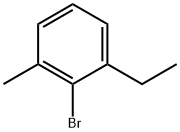
Purity:97%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
hydrogen bromide; copper(I) bromide; sodium nitrite; water;
Reactants are commercially availlable.
GLAXO GROUP LIMITED, WO2006/100119, A1, (2006) Description 3; - l-Bromo-2-ethyl-6-methylbenzene2-Ethyl-6-methylaniline (35 g, 0.259 mol) was added to cooled concentrated hydrobromic acid (120 mL). The stirred suspension was maintained between 5°C and 9°C while a solution of sodium nitrite (18.2 g, 0.264 mol) in water (40 mL) was added. A solutio n of copper (I) bromide (20.4 g, 0.142 mol) in concentrated hydrobromic acid (28 mL) was added and t he reaction mixture was allowed to rise to room temperature. The mixture was stirred at room tempera ture for 1 hour and then 30 minutes at 40°C. After cooling to room temperature hexane was added. The organic layer was washed with water, dried (MgSO4) and evaporated. Purification by chromatography on silica gel (hexane) gave the title compound. 1H NMR (CDCl3) delta 1.22 (t, 3H), 2.42 (s, 3H), 2.78 (q, 2H), 7.04-7.14 (m, 3H).
