Product name:4-Chloro-5,8-difluoro-2-methyl-quinoline
MF:C10H6ClF2N
Cas No.:288151-28-0
MW:213.61
Structure: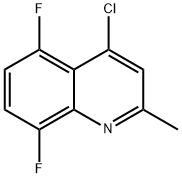
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
trichlorophosphate;
Reactants are commercially availlable.
SmithKline Beecham p.l.c., US6699879, B1, (2004) 4-Hydroxy-5,8-difluoro-2-methylquinoline (5.4 g) in phosphoryl chloride (60 ml) was boiled for 4 h. The mixture was cooled to room temperature, excess phosphoryl chloride removed at reduced pressure, the residue dissolved in ethyl acetate, washed with sodium hydrogen carbonate, dried (Na2SO4) and solvent removed at reduced pressure. The title compound (5.35 g) was isolated as a brown powder. (Meth od B). 1H NMR delta: 2.61 (3H, s), 7.46 (1H, m), 7.66 (1H, m), 7.81 (1H, s).
