Product name:7-bromopyrrolo[1,2-f][1,2,4]triazin-4-amine
MF:C6H5BrN4
Cas No.:937046-98-5
MW:213.03
Structure: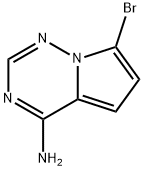
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
1,3-dibromo-5,5-dimethyl-hydantoin; N,N-dimethyl-formamide;
Reactants are commercially availlable.
BAYER PHARMACEUTICALS CORPORATION, WO2007/56170, A2, (2007) A stirred solution containing Intermediate A (21.0 g, 0.157 mol) in anhydrous DMF (200 ml_) was cooled to -20 0C and 1 ,3-dibromo-5,5-dimethylhydantoin (22.4 g, 0.078 mol) was added portionwise over - 45 minutes. The reaction was stirred for another 45 minutes and monitored for completion by TLC (silica gel, GHLF, 5percentCH3OH/CH2Cl2). Saturated25 Na2SO3 solution (300 mL) was added, the resulting suspension was stirred and the solids were collected by suction filtration. The filter cake was washed with water, dried by suction and then partitioned between ethyl acetate (1 L) and 5percent sodium carbonate solution (1L). The layers were separated, the organic layer was washed with fresh sodium carbonate solution and dried over magnesium sulfate. The filtrate from the work-30 up was also extra cted and combined with the main batch then filtered through a pad of Magnesol and concentrated in vacuo to afford crude mono-bromide, KRAM 206-3-1 , 29.9 g, 90percent yield. Trituration of a 21.5 g quantity of the crude product in hot ethyl acetate (300 mL, 70 C) provided colorless solids (12.3 g) containing only ~2 percent of the di-brominated side-product. 1H-NMR (CD3OD) : delta 7.84 ( s, 1 H), 6.95 (d, 1 H, J= 4.7 Hz), 6.71 (d, 1H, J = 4.7 Hz), 4.89 (s, 3H, -NH2 + H2O); MS: LC/MS (+esi), m/z = 213.1 [M+H].
