Product name:2-Methyl-5-pyrimidinamine
MF:C5H7N3
Cas No.:39889-94-6
MW:109.13
Structure: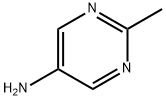
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
palladium 10 on activated carbon; hydrogen; sodium hydroxide; diethyl ether; water;
Reactants are commercially availlable.
MARKWALDER, Jay A., WO2014/150677, A1, (2014) Example 77 (1 S,2R)-2-(4-(diisobutylamino)-3-(3-(2-methylpyrimidin-5-yl)ureido)phenyl)cyclopropanecarboxylic acid 77A. 2-methylpyrimidine-5-amineA solution of 4,6-dichloro-2-methylpyrimidin-5-amine (2 g, 11.23 mmol) in ethylether (93 ml) was treated with sodium hydroxide (7.37 g, 184 mmol) in water (22.05 ml)and 10percent palladium on carbon (0.161 g, 1.5 17 mmol). The mixture was shaken at rt on aParr shaker under 50 psi of H2 gas for 22 h. The reaction was filtered through Celite andthe filter cake was washed with DCM. The solvent from the filtrate was evaporated togive a yellow residue. The suspension was re-dissolved in DCM and water. The aqueous layer was neutralized to approximately pH 6 with 4N HC1, then extracted with DCM (3X). The combined organic phases were dried over anhydrous Na2SO4, filtered, and concentrated to afford a yellow residue. The aqueous phase still contained product, so thewater was evaporated to give a yellow solid. The solid was taken up in MeOH and DCM and filtered to remove all salts. The filtrate was evaporated to give a yellow residue. A total of two crops were obtained - one from the extraction and one from the aqueous layer. Each crop was purified by flash chromatography and combined to give 77A (off- white solid, 0.968 g, 8.87 mmol, 79 percent yield). ?H NMR (400MHz, CHLOROFORM-d) oe8.14 (s, 2H), 3.60 (br. s., 2H), 2.61 (s, 3H).
