Product name:tert-butyl 2,6-dimethyl-4-oxopiperidine -1-carboxylate
MF:C12H21NO3
Cas No.:604010-24-4
MW:227.31
Structure: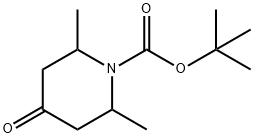
Purity:95%
Appearance:light yellow solid
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
triethylamine; dichloromethane;
Reactants can be synthesized in 1 step.
KESICKI, Edward A., WO2015/103060, A1, (2015) [0359] Preparation of tert-butyl 2,6-dimethyl-4-oxopiperidine-l-carboxylate (9.37): To an ice-cold solution of 2,6-dimethylpiperidin-4-one 9.36 (3.6 g, 28.34 mmol) in DCM (50 mL) was added Et3N (9.8 m L, 70.8 mmol) and boc anhydride (12.1 mL, 56.6 mmol). The resulting reaction mixture was stirred at RT for 16 h. After completion of the reaction (TLC monitoring), the reaction mass was diluted with w ater and extracted with DCM (3 times). The combined organics was washed with brine, dried over anhydrous Na2S04, filtered and concentrated. The crude was purified over silica gel (100-200 M, 10-15perc ent) EtOAc-hexane) to get the desired product 9.37 (3.9 g, Yield: 60.9percent). [0360] 1H-NMR (400 MHz, CDC13): delta 2.85 (d, J=6.4 Hz, 1H), 2.81 (d, J=6.4 Hz, 1H), 2.38-2.39 (m, 2H), 2.34-2.36 (m, 2 H), 1.48 (s, 9H) and 1.25-1.27 (m, 6H). MS: 228.08 (M+H)+.
