Product name:[2R-(2a,3b,11bb)]-1,3,4,6,7,11b-Hexahydro-9,10-dimethoxy-3-(2-methylpropyl)-2H-benzo[a]quinolizin-2-ol
MF:C19H29NO3
MW: 319.44
Cas No.:85081-18-1
Structure: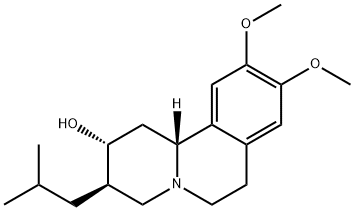
Purity:97%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
L-Selectridereg;; tetrahydrofuran; ethanol;
Reactants are commercially availlable.
TRIDGETT, Robert, WO2005/77946, A1, (2005) 1M L-SelectrideX in tetrahydrofuran (135 ml, 135 mmol, 2.87 eq. ) was added slowly over 30 minutes to a stirred solution oftetrabenazine RR/SSracemate (15 g, 47 mmol) in ethanol (75 ml) and tetrahydro furan (75 ml) at 0 °C. After addition was complete the mixture was stirred at 0 °C for 30 minutes an d then allowed to warm to room temperature. The mixture was poured onto crushed ice (300 g) and wate r (100 ml) added. The solution was extracted with diethyl ether (2 x 200 ml) and the combined ethereal extracts washed with water (100 ml) and partly dried over anhydrous potassium carbonate. Drying w as completed using anhydrous magnesium sulphate and, after filtration, the solvent was removed at re duced pressure (shielded from the light, bath temperature <20 °C) to afford a pale yellow solid. The solid was slurried with petroleum ether (30-40 °C) and filtered to afford a white powdery solid (12 g, 80percent).; 2A. Preparation of 2 3-Dehydrotetrabenazine; A solution containing a racemic mixtur e (15 g, 47 mmol) of RR and SS tetrabenazine enantiomers in tetrahydrofuran was subjected to reduction with L- Selectridee by the method of Example 1A to give a mixture of the 2S, 3R, 1 lbR and 2R, 3S , 1 lbS enantiomers of dihydrotetrabenazine. as a white powdery solid (12 g, 80percent). The partially purified dihydrotetrabenazine was then dehydrated using PCl5 according to the method of Example 1 B to give a semi-pure mixture of 1 lbR and 1 lbS isomers of 2, 3-dehydrotetrabenazine (the 1 lbR enantiomer of which is shown below) as a yellow solid (12.92 g, 68percent).
