Product name:2-Methylthiazol-5-amine
MF:C4H6N2S
Cas No.:89281-44-7
MW:114.17
Structure: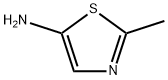
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
triethylamine; methanol; methanol;
Reactants can be synthesized in 2 steps.
ASTRAZENECA UK LIMITED, WO2007/71955, A1, (2007) Method 2; 2-Methgammal- 1 ,3-thiazol-5-amine; To a solution of aminoacetonitrile bisulfate (6.4 g, 41.6 mmol) in anhydrous MeOH (75 ml) at O0C was added Et3N (11.6 mL, 83mmol). After 30 minutes, ethyl dithioacetate (5g, 41.6 mmol) was added and the resulting dark orange solution stirred at room temperature for 2 hours. Half the solvent was removed under reduced pressure. The solution was diluted w ith an equivalent volume of EtOAc, washed with water, and dried (Na2SO4). The solvents were removed under reduced pressure and the residue slurried in warm EtOAc, cooled in an ice bath, and filtered t o give 2.45g (52percent) of a brown solid. 1H NMR DMSO-d6: 6.62 (s, 1 H) 5.36 (bs, 2 H) 2.89 (s, 3 H); m/z: 115.
