Product name:ETHYL 4,4-DIFLUOROCYCLOHEXANECARBOXYLATE
MF:C9H14F2O2
Cas No.:178312-47-5
MW:192.2
Structure: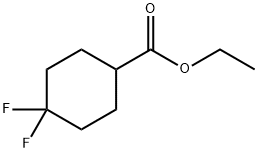
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
hydrogen fluoride;
Reactants are commercially availlable.
SOLVAY (Societe Anonyme), EP1985603, A1, (2008) The obtained mixture (63.7 g) of comparative example 1 was used for the fluorination using HF (157.6 g) in a PTFE flask. HF was then introduced at dry ice temperature. The mixture was stirred at room temperature overnight. Afterwards water was added and the occurring phases were separated. The crude was washed three times with 100 ml of water. 20.4 g were obtained and analyzed via GC. The reaction yield of ethyl 4,4-difluorocyclohexanoate was 25percent. The above examples and comparative examples demonstrate inter alia that the process according to the present invention affords the desired pro duct with higher yields than the prior art processes.
