Product name:METHYL 2-FLUOROACRYLATE
MF:C4H5FO2
Cas No.:2343-89-7
MW:104.08
Structure: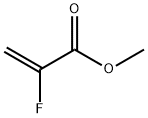
Purity:98%
Appearance:colorless liquid
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
2,6-di-tert-butyl-4-methyl-phenol; sodium salt of phosphorous acid; 1-methyl-pyrrolidin-2-one;
Reactants are commercially availlable.
SALTIGO GMBH, US2012/283468, A1, (2012) 2. Preparation of methyl 2-fluoroacrylateAn amount of di-tert-butylhydroxytoluene (BHT) of 4 g (0.02 mol), 500 g of N-methylpyrrolidine (NMP) and 360 g (2.2 mol) of tribasic sodium phosphate are intro duced into a sulfonation pan and the mixture is heated to 150°C. An amount of di-tert-butylhydroxyt oluene (BHT) of 51 mg (2.2*10-4 mol) is introduced into the distillation receiver of the vertical re covery bend. A vacuum of 300 mbar is applied and the metered addition of a total of 287 g (98percent, 2.0 mol) of methyl 3-chloro-2-fluoropropionate is begun. The metered addition is continued in the degree in which the methyl 2-fluoroacrylate is distilled over. When no more distillate stream passes over, the vacuum is lowered to 150 mbar. When no more product passes over even here, the pressure i s raised by addition of nitrogen. The distillation bottoms are cooled and discharged via the bottom valve. The product is obtained after distillation as a colorless liquid (205 g, 95percent, 1.87 mol, 93.5percent yield).
