Product name:2-Amino-3,5-dihydro-pyrrolo[3,2-d]pyrimidin-4-one
MF:C6H6N4O
MW: 150.14
Cas No.:65996-58-9
Structure: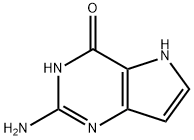
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
triethylamine; mercury dichloride; sodium acetate; sodium hydroxide; N,N-dimethyl-formamide; methanol; methanol; water;
Reactants are commercially availlable.
BLAGG, Julian, WO2011/35009, A1, (2011) Preparation of Intermediate DDTo a mixture of Intermediate CC (200 mg, 1.049 mmol), Et3N (0.436 mL, 3.14 mmol), and l ,3-dicarbomethoxy-2-methyl-2-thiopseudourea (216 mg, 1.049 mmol) in DMF (8 mL) was added HgCl2 (284 mg, 1.049 mmol) at room temperature (26 °C). The mixture was stirred overnight. Th e reaction mixture was then filtered, and the filtrate was diluted with H20 (50 mL), extracted with EtOAc (-150 mL), washed with H20 (50 mL) and brine solution (50 mL), dried (Na2SC>4), filtered, and evaporated to obtain crude Intermediate DD (150 mg). This material was used in the next step without any further characterization and purification. Rf = 0.3 (20percentEtO Ac/petroleum ether). Mass: (m z=313.1).Preparation of Compound (33)To a solution of Intermediate DD (150 mg, 0.48 mmol) in MeOH ( 10 mL) was added NaOMe (129 mg, 2.40 mmol) at 0 °C. The reaction was warmed to room temperature and stirred overnight. The reaction mixture was concentrated, and IN aqueous NaOH (2.5 mL) was added. T he reaction was heated to 60 °C for 30 minutes. The reaction mixture was evaporated to obtain a crud e residue that was purified by column chromatography (100-200 mesh silica gel, 20percent) MeOH/CHCl3 /aq. NH3) to obtain Compound (33) (15 mg, 9.6percent). Rf = 0.6 (20percent MeOH/CHCl3/0.1 mL aqueous NH3). 1H-NMR (400MHz, DMSO-J6) delta 11.36 and 1 1.20 (2 overlapped br. s, exchanged with D20, 2H), 7.05 (s, 1H), 6.05 (br. s, exchanged with D20, 2H), 5.88 (d, J = 2.4Hz, 1H). Mass (m/z): 150.7 (M++l). LCMS: (Column: Zodiacsil 120-5-C- 18-Aq (4.6 chi 50 mm), Mobile phase: A: 0.01M HCOONH4 (Aq); B: MeOH, T/percentB: 0/5, 10/90, 10.1/5, Flow: 1.0 mL/min, Diluent: MeOH), Rt=2.379 min, 97.08 (214 nm), 98.26 (254 nm).
