Product name:2-Chloropyridine-3-sulfonyl chloride
MF:C5H3Cl2NO2S
MW: 212.05
Cas No.:6684-06-6
Structure: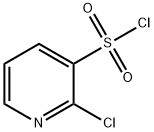
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
hydrogenchloride; sulfur dioxide; acetic acid; sodium nitrite;
Reactants are commercially availlable.
Ishihara Sangyo Kaisha Ltd., US5869428, A1, (1999) (1) 50.0 g of 3-amino-2-chloropyridine, 100 ml of hydrochloric acid and 40 ml of acetic acid were mixed, and 100 ml of an aqueous solution containing 29.5 g of sodium nitrite was dropwise added theret o at a temperature of not higher than 5°C. with stirring. The mixture was reacted at the same tempe rature for one hour. The obtained suspension was gradually added to 400 ml of acetic acid containin g 3 g of cupric chloride and 75 g of sulfur dioxide gas at 0°C. with stirring, followed by a reacti on at the same temperature for 30 minutes to obtain a reaction mixture containing 2-chloro-3-chloros ulfonylpyridine. Water was added to the obtained reaction mixture, and then the mixture was extracted with methylene chloride. Then, the extract layer was thoroughly washed with water and dried over a nhydrous sodium sulfate.
