Product name:6-Methylpyridine-3-sulfonyl chloride
MF:C6H6ClNO2S
Cas No.:478264-00-5
MW:191.64
Structure: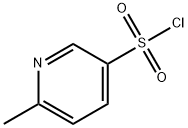
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
hydrogenchloride; sodium nitrite; hydrogenchloride; sodium hydrogensulfite; copper(II) sulfate; water; water;
Reactants are commercially availlable.
Takeda Pharmaceutical Company Limited, US2007/60623, A1, (2007) Reference Example 186 6-methylpyridin-3-ylsulfonyl chloride; To a mixture of 6-methylpyridine-3-amine (449 mg) and concentrated hydrochloric acid (5 mL) was added a solution of sodium nitrite (857 mg) in water (2 mL) at 0°C., and the mixture was stirred at the same temperature for 10 min. To the mi xture was added a solution of concentrated hydrochloric acid (2.5 mL), copper sulfate (69 mg) and so dium hydrogen sulfite (5.08 g) in water (8 mL) at 0°C., and the mixture was stirred at room tempera ture for 30 min. The reaction mixture was extracted with ethyl acetate. The extract was washed with saturated brine, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure. The residue was purified by silica gel column chromatography (eluent: hexane-ethyl acetate=10:1) to gi ve the title compound as a pale-yellow solid (yield 0.12 g, 15percent). 1H-NMR (CDCl3)delta: 2.73 (3H, s), 7.40-7.43 (1H, m), 8.16-8.20 (1H, m), 9.11-9.12 (1H, m).
