Product name:2-CARBOMETHOXY-3-TROPINONE
MF:C10H15NO3
Cas No.:36127-17-0
MW:197.23
Structure: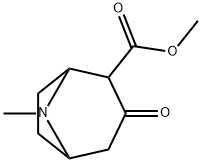
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
toluene;
Reactants can be synthesized in 1 step.
Nihon Medi-Physics Co., Ltd., US6486323, B1, (2002) EXAMPLE 5 Synthesis of Methyl 8-Methyl-3-oxo-8-azabicyclo[3.2.1]octane-2-carboxylate (2a: R'=Me and R=Me in the Formula (2)) Lipase PS (3.79 g) was added to a mixture of the dimethyl 8-methyl-3-oxo-8- azabicyclo[3.2.1]octane-2,4-dicarboxylate (1a) (430 mg, 1.68 mmol) of obtained in Example 1, toluene (10 ml) and 0.3 M phosphate buffer (pH=7.2, 10 ml), and the resulting mixture was stirred at room t emperature for 3 days. After completion of the reaction, the reaction mixture was heated at 80°C. for 10 minutes and filtered with Celite, and the filtrate was adjusted to pH 8-8.5 with a saturated aqueous sodium hydrogencarbonate solution and extracted with chloroform. The extract was dried over Na2SO4, filtered and concentrated in vacuo, and the thus obtained crude product was purified by a si lica gel column chromatography (CHCl3:MeOH=40:1) to obtain 161 mg of methyl 8-methyl-3-oxo-8-azabicyclo[3.2.1]-octane-2-carboxylate. The yield was 48.5 mol percent.
