Product name:Ethyl 2-aminothiazole-5-carboxylate
MF:C6H8N2O2S
Cas No.:32955-21-8
MW:138.19
Structure: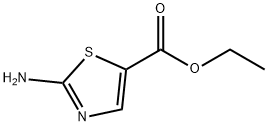
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
sodium; ethanol; ethanol;
Reactants are commercially availlable.
ELI LILLY AND COMPANY, WO2004/80996, A1, (2004) Reference : PLOUVIER, B. ; BAILLY9 C. ; -LOUSSIN, F.; HENICHART, J. P. Heterocycles, 1991, 32 (4) 69 3-701. Dissolve sodium (4.60 g, 200 mmol) in ethanol (100 mL). Cool the solution to-5 °C to-10 °C an d add a mixture of ethyl fonnate (16.8 mL, 220 mmol) and ethyl chloroacetate (27.0 mL, 220 mmol). A white precipitate forms. Add ether (200 mL) to facilitate the precipitation. Warm the reaction mixtu re to room temperature and stir over night. Filter the precipitate and wash it with ether. Concentrate the filtrate to ABOUT 1/4 OF the volume, add ether (100 mL) and refilter. Dissolve the combined f ilter cakes with 10percent HCl (100 mL). Extract the acidic aqueous phase with ether (3 X 100 mL). Dry the organic layer over NA2SO4, filter and concentrate. Dilute the liquid with ethanol (106 mL) an d add THIOUREA (8.07 g 106 mmol). Heat the reaction mixture to 80 °C. After 1.25 hours, concentrate the reaction mixture. Then take the dark oil up in 1.0 N NAOH (100 ML) and extract with dichlorometh ane (3 X 100 mL). Dry the organic extracts over NA2SO4, filter and concentrate. Suspend the solid with methanol, then add chloroform. Filter the resulting precipitate as the title compound: TOF MS ES+ 173.0 (M+H) +, HRMS calcd for C6H9N202S 173.0385 (M+H) +, found 173.0381, time 0.36 min; HPLC [YMC-Pro pack C-18 (150 x 4.6 mm, S-5 microm), 0.05percent TFA/acetonitrile in 0.05percent TFA/WATER at 1 .0 ML/MIN, 10-20percent over 5 min, 20-95percent over 18], tR = 6.2 min, 100percent purity.
