Product name:3,6-Dihydrothiopyran-4-Boronic Acid Pinacol Ester
MF:C11H19BO2S
Cas No.:862129-81-5
MW:226.14
Structure: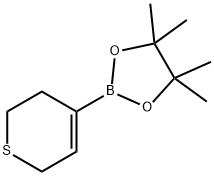
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
potassium phenolate; toluene; bis-triphenylphosphine-palladium(II) chloride; triphenylphosphine
Uncertain vulnerable functions: sulfide.
Reactants can be synthesized in 1 step.
AMGEN INC., WO2009/111056, A1, (2009) Synthesis of T12.2. A mixture of triphenylphosphine (0.90 g, 3.4 mmol), potassium phenolate (6.4 g, 48 mmol), 4,4,5,5-tetramethyl-2-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1,3,2-dioxaborolane (8.7 g, 34 mmol) and T12.1 (8.00 g, 34 mm ol) in toluene (172 mL) was degassed by N2. Then dichlorobis(triphenylphosphine)palladium(II) (1.2 g, 1.7 mmol) was added, and the reaction mixture was further degassed with N2. The reaction was then stirred at 50°C for 3.5 hours. The react ion was then filtered and concentrated in vacuo. The product was purified on silica gel (0-5percent EtOAc in hexanes) to yield T12.2 as a colorless oil (5.0 g, 69percent yield).
