Product name:3,6-Dihydro-2H-pyran-4-boronic acid pinacol ester
MF:C11H19BO3
Cas No.:287944-16-5
MW:210.08
Structure: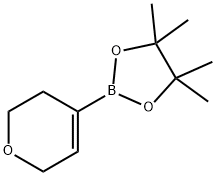
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
(1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium acetate; 1,4-dioxane;
Reactants are commercially availlable.
Under an atmosphere of nitrogen, a solution of 4-bromo-3,6-dihydro-2H-pyran 10 (24.5 g, 0.15 mol), bis(pinacolato)diboron (42.0 g, 0.15 mol),1,1?-bis(diphenylphosphino)ferrocene-palladium(II) dichlori de methylene chloride complex (1.25 g, 1.5mmol) and potassium acetate (44.5 g, 0.45 mol) in dioxane (350 mL) was heated to 80 oC for 3 h. Water (50 mL) was added to the reaction mixture which then was extracted with CH2Cl2 (3 x 30 mL). The combined organic extracts were washed with brine, dried over anhydrous sodium sulfate and evaporated under reduced pressure. The residue was recrystallized in cold hexane to yield the title compound 1 (20.6g, 0.098 mol, 66percent yield) as white solid. mp 61?6 2 °C (lit.11 60?61 °C); 1H NMR (400 MHz, CDCl3): delta6.53 (s, 1H), 4.20?4.18 (m, 2H), 3.77?3.74 (t, J = 6 Hz, 2H), 2.25?2.23 (m, 2H), 1.27 (s, 12H); HRMScalcd for C11H19BO3 [M]+ 210.1427; found: 210.1429.
