Product name:2-Amino-5-methyl-3-thiophenecarbonitrile
MF:C6H6N2S
Cas No.:138564-58-6
MW:138.19
Structure: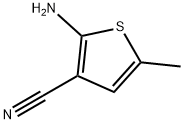
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
sulfur; triethylamine; N,N-dimethyl-formamide; N,N-dimethyl-formamide;
Reactants are commercially availlable.
RAMOT AT TEL-AVIV UNIVERSITY LTD, WO2008/50341, A2, (2008) For a general synthesis see He, X.; Griesser, U. J.; Stowell, J. G.; Borchrdt, T. B.; Byrn, S. R. J. Pharm. Sci. 2001, 90, 371.A Sulfur (S8, 0.9 g, 0.0 27 mmol), propionalaldehyde (2 ml, 0.027 mmol) and DMF (6 ml) were transferred to a thre e-necked round-bottomed flask equipped with a dropping funnel and condenser. The resulting mixture was cooled to 0°C, and EPO triethylamine (2.3 ml) was added in a dropwise manner via the dropping funnel. The resulting dark sol ution was then warmed to room temperature over a period of 1 h. A solution of malononitrile (1.71 ml, 1.8 g, 0.027 mmol) in DMF (3.2 ml) was transferred to the addition funnel and added in a dropwise manner. The resulting brownish mixture w as stirred overnight at room temperature. Then the mixture was poured over 80 ml of ice and water to yield an orange precipitate. The solid A was filtered, washed with chilled water, and dried in vacuo; yield 78percent.1H NMR (200 MHz, CDCl 3): delta 6.35 (s, IH), 4.15 (br s, 2H), 2.27 (s, 3H). 13C NMR (50 MHz, CDCl3): delta 160.9, 146.3, 124.6, 122.0, 115.7, 14.9.
