Product name:(R)-tert-butyl 2-methyl-4-oxopiperidine-1-carboxylate
MF:C11H19NO3
MW: 213.27
Cas No.:790667-43-5
Structure: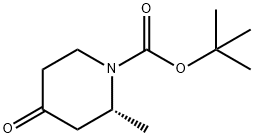
Purity:97%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
toluene-4-sulfonic acid; acetone;
Reactants are commercially availlable.
Sankyo Company, Limited, EP1375482, A1, (2004) To a solution of 1-t-butoxycarbonyl-2-methyl-4-piperidone ethylene ketal (6.00 g) obtained in reference example 82 in acetone (150 ml) was added p-toluenensulfonic acid monohydrate (4.40 g) with stirr ing under ice-cooling, and the resulting mixture was stirred at room temperature overnight. After stirring, the reaction mixture was diluted with ethyl acetate, and the organic layer was washed succes sively with a saturated sodium hydrogencarbonate solution and a saturated aqueous sodium chloride solution. The organic layer was dried over anhydrous magnesium sulfate and evaporated in vacuo to affo rd the title compound (2.40 g, yield: 48 percent) as a yellow oil. 1H NMR (500MHz, CDCl3) delta ppm : 1.18 (3H, d, J=7.0), 1.49 (9H, s), 2.20-2.30 (1H, m), 2.30-2.40 (1H, m), 2.45-2.55 (1H, m), 2.6 5-2.70 (1H, m), 3.25-3.35 (1H, m), 3.90-4.05 (1H, m), 4.20-4.30 (1H, m).
