Product name:Tert-butyl 5-(HydroxyMethyl)thiazol-2-ylcarbaMate
MF:C9H14N2O3S
Cas No.:1001419-37-9
MW:230.28
Structure: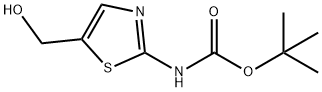
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
lithium aluminium tetrahydride; sodium hydroxide; water; tetrahydrofuran; tetrahydrofuran;
Reactants are commercially availlable.
Zhong, Min, US2006/35908, A1, (2006) Step 1: To a suspension of 2-tert-butoxycarbonylamino-thiazole-5-carboxylic acid methyl ester (compound 62.1; 21.7 mmol) in THF was slowly added LAH (25 mL of a 1.0 M solution in THF). After stirring for 1 hour at room temperature the reaction is cautiously hydrolyzed with the sequential addition of water (0.95 mL), 15percent aqueous NaOH (0.95 mL) and additional water (2.85 mL). After stirring fo r 30 minutes the reaction is filtered through a pad of Celite and the filtrate is dried and concentrated to give (5-hydroxymethyl-thiazol-2-yl)-carbamic acid tert-butyl ester (compound 62.2).
