Product name:2-Amino-5-bromothiazole monohydrobromide
MF:C3H4Br2N2S
Cas No.:61296-22-8
MW:259.95
Structure: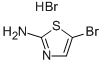
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
bromine; chloroform; AlCl3, aluminium chloride
Reactants are commercially availlable.
Wyeth, US2007/4786, A1, (2007) EXAMPLE 112; Preparation of 2-Amino-5-(3bromophenyl)-5-[5-(2-ethyl-[1,3]dioxolan-2-yl)-thiophen-3-yl]-3-methyl-3,5-dihydro-imidazol-4-one; Step a); A stirred suspension of aluminum trichloride (6.27 g, 47.1 mmol) in chloroform at room tempe rature was treated sequentially with 2-(1-propionyl)thiophene (3.00 g, 21.4 mmol) and bromine (3.60 g, 22.4 mmol), stirred for 17 h, poured into iced water and diluted with methylene chloride. The phases were separated and the aqueous phase was extracted with methylene chloride. The extracts and the organic phase were combined, washed with water and brine, dried over sodium sulfate and concentrated to afford 2 (5.51 g, 117percent) as a dark orange oil: 1H NMR (300 MHz, CDCl3) delta 7.59 (d, J=1.1 Hz, 1H), 7.51 (d, J=1.1 Hz, 1H), 2.91 (q, J=7.3 Hz, 2H), 1.23 (t, J=7.3 Hz, 3H); ESI MS m/z 219 [C7H7BrOS+H]+.
