Product name:(5-methylpyridin-2-yl)methanol
MF:C7H9NO
MW: 123.16
Cas No.:22940-71-2
Structure: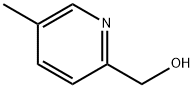
Appearance: yellow liquid
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
acetic anhydride; hydrogenchloride; water;
Reactants can be synthesized in 1 step.
GALLASCHUN, Randall, WO2014/143799, A2, (2014) Step 2 (5-methylpyridin-2-yl)methanol 2, 5-Dimethylpyridine 1 -oxide (2.5g, 20.3 mmol) was added to acetic anhydride (8.2mL) at 100 °C over a period of 30 min. Then the mixture was refluxed 1 h. The m ixture was cooled to room temperature and carefully quenched with ethanol (1 lmL). The reaction mixture was concentrated. The residue was treated with IN HC1 (6 mL) and refluxed 1 h. The reaction was concentrated and the residue was partitioned between ethyl acetate and water. The organic phase was dried over sodium sulfate, filtered and concentrated to give (5-methylpyridin-2-yl) methanol (2g, 80 .0percent yield) as a light yellow oil. LCMS MH+ 124.
