Product name:2-BROMO-4-HYDROXYMETHYLTHIAZOLE
MF:C4H4BrNOS
Cas No.:5198-86-7
MW:194.05
Structure: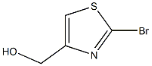

Purity:95%
Appearance:brown liquid
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
sodium tetrahydroborate; water; lithium chloride; tetrahydrofuran;
Reactants are commercially availlable.
PETERSON,Michael, T., WO2015/57585, A1, (2015) Preparation of (2-bromothiazol-4-yl)methanol (5)To a solution of ester 4 (10.0 g, 42.4 mmol, 1 equiv) in THF (200 mL) was added NaBH4 (4.81 g, 127 mmol, 3 equiv), LiCl (5.4 g, 130 mmol, 3 equiv) and H2O (40 mL). The resulting biphasic mixtu re was vigorously stirred for 2 h, after which TLC (20percent EtOAc:hexanes) showed complete consumption of starting material. The reaction was quenched with saturated aqueous NH4Cl (200 mL), diluted with EtOAc (200 mL), and the layers were separated. The aqueous layer was extracted with EtOAc (100 mL), and the organic layers were combined, dried (Na2SO4), filtered, and concentrated under reduced pressure to give the title compound as a yellow semi-solid (7.82 g, 95percent). Rf= 0.2 (SiO2, 20percent EtOAcrhexanes); 1H and 13C NMR of the product matched those previously reported (Wipf and Wang 2007).
