Product name:3-Cyano-5-fluorobenzeneboronic acid pinacol ester
MF:C13H15BFNO2
Cas No.:935685-88-4
MW:247.0731032
Structure: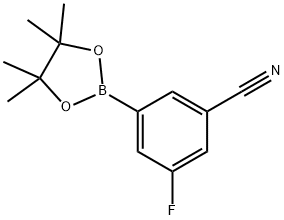
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
potassium acetate; 1,4-dioxane; dichloromethane; dimethyl sulfoxide; dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II) dichloromethane adduct
Reactants are commercially availlable.
AiCuris GmbH and Co. KG, US2012/22123, A1, (2012) Example 5A3-Fluoro-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzenecarbonitrileUnder argon, 3.60 g (18.0 mmol) of 3-bromo-5-fluorobenzenecarbonitrile, 5.03 g (19.8 mmol) of 4,4,4',4',5,5,5',5'-o ctamethyl-2,2'-bi-1,3,2-dioxaborolane and 5.30 g (54.0 mmol) of potassium acetate are provided in 72 ml of degassed 1,4-dioxane/DMSO (10/1), and 441 mg (0.54 mmol) of 1,1'-bis(diphenylphosphine)ferroc enedichloropalladium(II)/dichloromethane complex are added.The mixture is stirred at 90°C. overnigh t.Water is subsequently added, the phases are separated, the aqueous phase is extracted with ethyl a cetate and the combined organic phases are concentrated.The crude product is purified by flash chromatography (mobile phase: cyclohexane/ethyl acetate 10:1).4.48 g (92percent of theory) of the title c ompound are obtained.1H-NMR (400 MHz, DMSO-d6): delta=8.01 (ddd, 1H), 7.82 (s, 1H), 7.70 (ddd, 1H), 1.32 (s, 12H).GC-MS (Method 11): Rt=4.94 min; MS (EIpos): m/z=247 [M]+.
