Product name:4-AMINO-PIPERIDINE-1-CARBOXYLIC ACID BENZYL ESTER
MF:C13H18N2O2
Cas No.:120278-07-1
MW:234.29
Structure: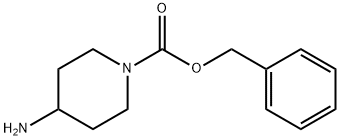
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
sodium iodide; N-methyl-2-(methylamino)ethylamine; ammonia; sulfolane; sulfolane; water; pentane; copper(l) iodide
Reactants are commercially availlable.
MASSACHUSETTS INSTITUTE OF TECHNOLOGY, WO2004/13094, A2, (2004) A 50 mL Schlenk tube was charged with CuI (385 mg, 2.02 mmol, 5.0 molpercent), NaI (9.00 g, 60.0 mmol), evacuated and backfilled with argon. N, N'-Dimethylethylenediamine (426 [1L,] 4.00 mmol, 10 molp ercent), sulfolan (20 mL), and [1-BROMO-2-METHYLPROPENE] (4.10 mL, 40.0 mmol) were added under argon . The Schlenk tube was sealed with a Teflon valve and the reaction mixture was stirred at [90 °C] fo r 24 h. The resulting dark green suspension was allowed to reach room temperature, poured into pentane (100 mL) and washed with a solution of 30percent aq ammonia (10 mL) in water (100 mL), followed b y water [(3X100] mL). The combined organic phases were dried [(MGS04)] and concentrated [TO No.10 ] mL volume. The residue was distilled collecting the fraction boiling at 120-124 [°C] to give 4. 82 g (66percent yield) of [L-IODO-2-METHYLPROPENE] as a colorless liquid.
