Product name:5-CHLORO-2-IODOPYRIDINE
MF:C5H3ClIN
MW: 239.44
Cas No.:244221-57-6
Structure: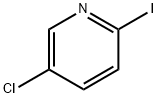
Appearance: Yellow solid
Purity:96%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
sodium iodide; acetonitrile;
Reactants are commercially availlable.
Ollis, Kristin P., US2003/45546, A1, (2003) a) 5-Chloro-2-iodo-pyridine To a refluxing solution of 2,5-dichloro-pyridine (12.2 g, 82.2 mmol), sodium iodide (37.0 g, 247 mmol) and acetonitrile (170 mL) under argon was added acetyl chloride (9. 0 mL, 127 mmol) and the solution was refluxed for 5 h. To the solution was added more sodium iodide (24.9 g, 166 mmol) and the solution was refluxed for 16 h. More sodium iodide (12.5 g, 83.6 mmol) wa s added and the solution was refluxed for 4.5 h. The solution was cooled to room temperature, and was partitioned between 10percent sodium thiosulfate/10percent aqueous sodium carbonate (300 mL) and e ther (400 mL). The ether layer was washed with brine (100 mL) and dried over anhydrous sodium sulfate. The solution was decanted, and dried in vacuo to yield 16.8 g (85percent) of the title compound a s a brown solid. 1H NMR (CDCl3): 8.36 (d, J=2.75 Hz, 1H), 7.66 (d, J=8.79 Hz, 1H), 7.32 (dd, J=8.38, 2.61 Hz, 1H). There is 5 mol percent of starting material 2,5-dichloro-pyridine in the product as d etermined by NMR.
