Product name:4-Methoxy-3-nitropyridine
MF:C6H6N2O3
MW: 154.12
Cas No.:31872-62-5
Structure: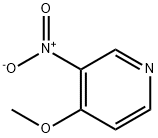
Appearance: Yellow solid
Purity:98%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
sulfuric acid; nitric acid; potassium carbonate;
Reactants are commercially availlable.
ZHAO, Zhicheng, WO2011/78984, A1, (2011) To cone. H2SO4 (5 mL) chilled in an ice bathm is added 4-methoxypyridine (0.5 mL, 4.9 mmol) dropwise over a 20 s period. Cone. Fuming nitric acid (5 mL) is added, and the reaction mixture is heated at 70 °C for 2.5 days. This mixture is cooled to rt, and then is poured into ice. Soild K2CO3 is added until the pH of the mixture is basic. The mixture is partitioned between H20 and EtOAc. The two lay ers is separated, and the aqueous layer is extracted with EtOAc once. The combined organic layers are washed with H2O and brine, dried over MgS04, filtered, and concentrated in vacuo to yield 0.7 g (9 2percent) of the product a yellow powder. 1H NMR (CDC13, 300 MHz) delta 9.02 (s, 1H), 8.65 (d, J= 5.8, 1 H), 7.04 (d, J = 5.9, 1H), 4.05 (s, 3H). LC Rt: 0.5 min; LCMS m/z 155 (M+l, 100percent).
