Product name:2,4-DICHLORO-1,3,5-TRIAZINE
MF:C3HCl2N3
Cas No.:2831-66-5
MW:149.97
Structure: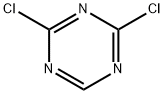
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthetic route is as follows:
trichlorophosphate; dichloromethane;
Reactants are commercially availlable.
VERTEX PHARMACEUTICALS INCORPORATED, WO2004/72063, A1, (2004) Example 9 2, 4-DICHLORO-1, 3, 5-triazine : Sodium DICYANAMIDE (LOG, 0.12 mol) is dissolved in water and added quickly to concentrated hydrochloric acid (60 mL) cooled to ABOUT-30 C. The slurry was sti rred at that temperature for about 15 minutes and then warmed to 35 C for 5 minutes before being cooled to 4 C for 45 minutes. The white precipitate was then filtered, washed with small amounts of wat er, dried under vacuum for 24 hours. About 5G of N-cyanochloro- FORMAMIDINE was obtained : 1H NMR (DMSO-D6): 8 7.59 (s, 1H). To a solution of DMF (1.1 equivalent, 6.0 ML, 77 mmol) in dichloromethane, at room temperature, was added phosphorous oxychloride (1.0 equivalent, 6.5 ML, 70 mmol) and then, after about 10 minutes, 1.0 eq. OF N-CYANOCHLOROFORMAMIDINE (6.25g, 70 mmol) was added. The mixture w as stirred overnight at room temperature and then washed 3 times with water and once with brine. The organic phase was dried over sodium sulfate, filtered, and evaporated under reduced pressure. The w hite solid (~4G) thus obtained wais identified as the 2, 4-DICHLORO-1, 3,5-triazine : LH NMR (CDC13) 6 8.88 (s, 1H).
