Product name:2-Bromo-6-hydroxypyridine
MF:C5H4BrNO
Cas No.:27992-32-1
MW:174
Structure: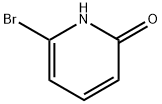
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
hydrogen bromide; dichloromethane;
Reactants are commercially availlable.
2-bromo-3-methoxypyridine (295 mg, 1.57 mmol) was heated in hydrobromide acid (5 ml) to 140 °C and s tirred overnight. The solution was neutralized with a saturated sodium bicarbonate solution and extr acted three times with ethyl acetate. The combined organic layers were dried over Na2SO4 and the solvent was removed under reduced pressure. Yield: 258 mg (94 percent), white crystals. 1H-NMR((CD3)2CO , 500 MHz): deltaH = 9.26 (s, 1H), 7.89 (dd, J = 1.6, 4.4 Hz, 1H), 7.32?7.36 (m,1H), 7.23?7.27 (m, 1H); 13C-NMR ((CD3)2CO, 125 MHz): deltaC = 152.4, 142.1, 131.9, 125.3, 124.5.
