Product name:4-BROMO-2-METHYLBENZYLAMINE
MF:C8H10BrN
MW: 200.08
Cas No.:376646-62-7
Structure: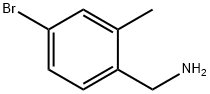
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
borane; hydrochlorid acid; tetrahydrofuran; 1,4-dioxane; ethyl acetate;
Reactants are commercially availlable.
MIAO, Hua, WO2015/89337, A1, (2015) [0617j To a solution of 4-bromo-2-methylbenzonitrile (3.0 g, 15 mmol) in anhydrous THF (20 mL) under nitrogen at 0 °C was added 1.0 M solution of borane in THF (46 mL). The reaction mixture was stirre d at 0 °C for 1 h, and heated at 80 °C overnight. The reaction mixture was cooled to 0 °C and slowly quenched with MeOH, concentrated in vacuo. The crude product was treated with EtOAc (20 mL) and 4 M of HC1 in 1,4-dioxane (8.0 mL, 32 mmol) for 5 mm. The solid was filtered, rinsed with diethyl ether, dried to give the title compound as a white powder (3.24 g, yield: 100percent). LCMS: RT 0.75 mm.; MH+ 200.0. ?H NMR (300 MHz, DMSOd6) 5: 8.28 (br. s., 2H), 7.42 - 7.54 (m, 2H), 7.34 (d, J = 7.93 Hz, 1H), 3.99 (d, J = 4.15 Hz, 2H), 2.35 (s, 3H).
