Product name:4,5-DIMETHYL-1,3-THIAZOL-2-AMINE
MF:C5H8N2S
Cas No.:2289-75-0
MW:128.2
Structure: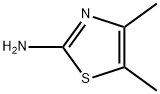
Purity:95%
Appearance:brown solid
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
n-butyllithium; tetrahydrofuran; tetrahydrofuran;
Reactants are commercially availlable.
Eisai RandD Management Co., Ltd., US2011/86882, A1, (2011) Production Example 20-12,5-Dimethyl-1,3-thiazole To a tetrahydrofuran (30 mL) solution of 2-methylth iazole (1.00 g, 10.1 mmol) was added n-butyllithium (4.02 mL, 10.6 mmol) dropwise at -78°C. and the mixture was stirred for 30 minutes. To the mixture was added iodomethane (0.69 mL, 11.1 mmol) and the mixture was warmed to room temperature. After the reaction was completed, water was added to the reaction mixture while cooling on ice, which was extracted with ethyl acetate. The organic layer was washed with brine and dried over anhydrous magnesium sulfate. The mixture was filtered to yield a f iltrate, and distilled off under reduced pressure to obtain the title compound (864 mg, 7.63 mmol).1H-NMR (CDCl3) delta: 2.41 (s, 3H), 2.64 (s, 3H), 7.24 (s, 1H).
