Product name:2-METHOXYACETAMIDINE HYDROCHLORIDE
MF:C3H9ClN2O
Cas No.:1903-91-9
MW:124.57
Structure: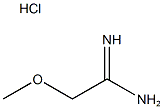
Purity:95%
Product use:Basic raw materials, organic intermediates, pharmaceutical intermediates
The synthesis process is as follows:
AlCl3, aluminium chloride; trimethylaluminum; silica gel; hydrochlorid acid; toluene; dichloromethane; toluene; methanol; diethyl ether;
Reactants are commercially availlable.
T illey, Jefferson Wright, US2009/264445, A1, (2009) Example 112 (S)-2-[4-(2-Chloro-phenoxy)-2-oxo-2,5-dihydro-pyrrol-1-yl]-4-methyl-pentanoic acid(3-methoxymethyl-[1,2,4]thiadiazol-5-yl)-amide To a stirred suspension of aluminum chloride (4.81 g, 0.090 mol) in toluene (33.5 mL) at 0°C. under argon was slowly added a solution of 2M trimethylaluminum in toluene (42 mL, 0.030 mol) maintaining a temperature below 10°C. The resulting mixture was allow ed to stir at room temperature for 2 h before a solution of 3-methoxyacetonitrile (5.56 g, 0.050 mol) in toluene (16.5 mL) was added. A fine light yellow precipitate formed and the resulting mixture w as warmed to 80°C. and allowed to stand for 20 h. The reaction mixture was cooled to room temperatu re and then slowly poured on to a cooled suspension of silica gel (40 g) in dichloromethane (100 mL) . The resulting mixture was stirred for 15 min, filtered and the silica gel pad washed well with methanol. The filtrate was evaporated and the residue was redissolved in a solution of dichloromethane/ methanol, filtered and evaporated which afforded a solid. The solid was suspended in a 3N hydrogen chloride solution in methanol (20 mL) and stirred vigorously while diethyl ether (500 mL) was added d ropwise. The resulting mixture was stirred at room temperature (30 min) decanted, washed with diethyl ether (250 mL) decanted and dried under vacuum which afforded 2-methoxy-acetamidine hydrochloride (4.66 g, 75percent) as a light yellow semisolid.
